Table of Contents
Africa carries 26% of the global disease burden, yet produces only about 3% of the world’s medicines. Over 70% of drugs used in Africa are imported. More than 95% of active pharmaceutical ingredients (APIs) also come from abroad, mostly from India and China.
That matters because it means “local manufacturing” often still depends on imported chemistry, imported logistics and imported pricing power. This heavy reliance on imports makes Africa vulnerable to global price changes and supply chain disruptions. There is a new, massive push of investment capital aimed at building local production capacity and reducing this risk.
The business potential is large enough to support this effort. Experts at Grand View Research value Africa’s drug market at $27.65 billion in 2024. They expect it to reach $36.96 billion by 2033, with non-branded generics growing the fastest at 4.4% per year.
The current investment trend is not just talk about being self-sufficient. It is a race to own drug factories in a large and growing market. However, Africa has seen many factory plans before. Simply building a pharmaceutical plant does not guarantee a successful business. The most viable plants are those with guaranteed buyers, clear legal approvals, and a plan to rely less on imported chemical ingredients over time.
Which Companies Are Building Africa’s New Pharma Plants?
Aspen is currently the best example of what a successful African pharma investment looks like. In 2024, the IFC, Proparco, DEG, and U.S. DFC gave Aspen a €500 million financing package to support Aspen’s “Manufacturing in Africa for Africa” strategy. This money supports making insulin, pediatric vaccines and broader medicine access for African patients. This followed another €600 million package in 2021 to grow Aspen’s vaccine manufacturing in South Africa. Altogether, Aspen has attracted over €1.1 billion from global lenders. This funding is geared towards real, existing medical needs and partnerships with international companies, not just a future plan.
Aspen is not building its manufacturing capacity based on speculative demand. Its deal with Novo Nordisk uses Aspen's sterile facility in Gqeberha to turn insulin into finished-dose vials for the African market. Aspen said this partnership aims to reach over 1 million patients in 2024, increasing to more than 4 million by 2026. Novo Nordisk expects 4.1 million people across Africa to be reached by 2026. This shows clear demand: named partners, specific products, existing facilities, and sales targets tied to a commercial deal, not just a general “Africa demand story.”
Aspen’s 10-year deal with the Serum Institute of India proves the same point for vaccines. The agreement covers four Aspen-branded routine vaccines such as pneumococcal, rotavirus, polyvalent meningococcal, and hexavalent using a technical-transfer and fill-finish process. Sales will go through African governments and multilateral organizations. The business plan is not just that vaccines are needed, it is that production is linked to clear purchasing channels. This is a much stronger setup than building factories first and hoping public buyers appear later.
The €50 million Fosun project near Abidjan serves as a clear example of growth in West Africa. IFC funding is helping build a factory in Côte d’Ivoire that can make 5 billion tablets a year. It will focus on antimalaria and antibacterial drugs for the region. The project’s strength relies on its massive scale and focus on needed drugs. However, it faces a big risk: these tablets must be cheaper than imports. It also operates in a region where regulatory fragmentation and procurement inconsistency remain real obstacles. While the new pharmaceutical plant manufactures drugs closer to patients, it still has to be profitable.
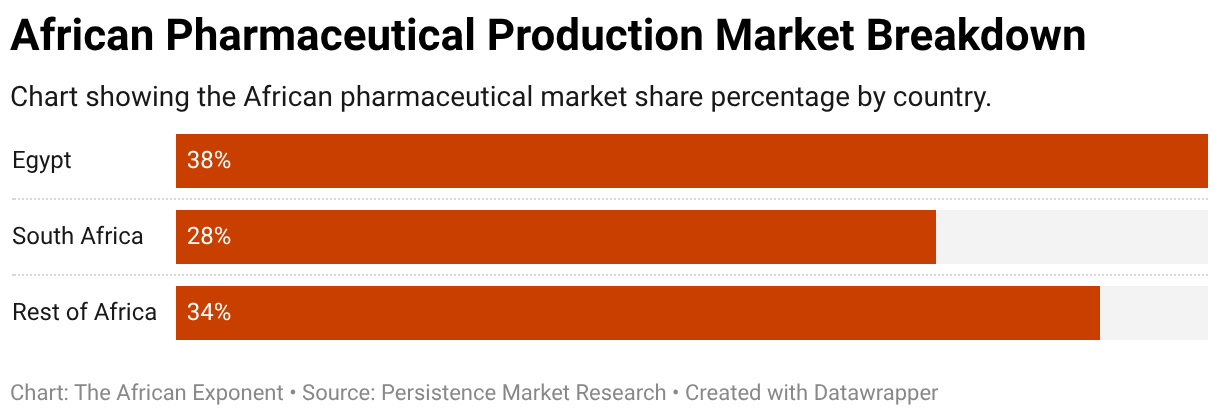
Egypt's Eva Pharma and Eli Lilly partnered to locally manufacture insulin glargine after gaining regulatory approval. Lilly supplies discounted API and technology transfer to help Eva Pharma formulate, fill and finish the insulin locally. This partnership aims to reach at least one million people annually by 2030 across 56 low- to-middle-income countries, mainly in Africa. In 2024, the model was extended to license the manufacturing of baricitinib (Olumiant) for 49 African countries, with supply expected from 2026. Eva's advantage is moving beyond simple packaging to higher-value licensed manufacturing and utilizing Egypt’s 38% market share and established export routes.
Nigeria is the most important swing market in the continent because of size, not because it has solved the model. The IFC notes the country imports about 70% of medical products but aims to produce 70% locally within five years. This ambition relies on upstream investment. Emzor's €14 million EIB-backed API project for antimalarials is important because it addresses local active-ingredient production which has been a neglected part of the value chain. If local API production remains rare, Nigeria will increase factory capacity without reducing its dependence on imported inputs.
What Determines Success or Failure for African Drug Manufacturing Plants?
The first test is demand. Aspen’s CEO, Stephen Saad, spoke clearly when discussing possible mpox vaccine manufacturing in the continent. Aspen needs both certain orders and funding for technology transfer. This is a lesson many African manufacturing projects have learned too late. Capital can finance the building of plants and equipment procurement, but it cannot keep empty factories running. The difference between a working plant and a failed one is usually not engineering. It is whether someone has already promised to buy the products.
The second factor is regulation. South Africa has a major lead because its regulator, SAHPRA, joined the International Council for Harmonisation (ICH) in December 2025. This alignment with global technical standards makes South African products more credible for export and attractive to international partners. In contrast, most other African nations require slow, country-by-country approvals that increase costs and delay new medicines.
The third factor is cost structure. Currently, Africa imports over 95% of its active pharmaceutical ingredients (APIs). This means even "local" factories depend on foreign suppliers for the most critical part of the drug manufacture. While Nigeria aims to produce 70% of its medical supplies locally by 2030, projects like Emzor’s €14 million API plant show that the continent is still in the early stages of building this essential chemical capacity. Without more local API production, African manufacturers will struggle to compete with cheaper Asian imports.
Is The Promised Regulatory Unlock Happening Fast Enough?
The African Medicines Agency (AMA) is vital for regulatory unlocking within the continent. As of January 2026, 31 member states had ratified the treaty establishing AMA. The African Union also appointed Dr Delese Mimi Darko as its first Director General in June 2025. This agency is important because it can simplify rules. If one approval works for many countries, Africa’s pharma industry becomes much more profitable than dealing with many different national regulators.
Investors should not confuse this promising direction with delivery. The Continental Listing pilot is a mechanism designed to test joint assessment procedures for medicinal products to create a unified regulatory pathway across Africa. While it is a good start, it is not yet a simple, one-stop system for getting drugs approved across the continent. Aspen’s Group Senior Executive Stavros Nicolaou's six-year qualification critique remains the operating reality for drug manufacturers deciding where to build today. Manufacturers still face long timelines to qualify their products.
For now, the best options are countries with strong national regulators. South Africa leads the way, especially after its regulator, SAHPRA, joined the ICH in December 2025. This move proves South Africa is the most reliable place on the continent for pharmaceutical investment thanks to its established global standards.
What Is The Investment Outlook For Africa’s Pharma Manufacturing Buildout?
The right way to read Africa’s pharma buildout is not as a story of self-sufficiency. Africa’s pharma growth is a mix of different strategies. Aspen is the strongest because it combines DFI capital, guaranteed multinational offtake, sterile-manufacturing capability and South Africa’s high regulatory standards. Eva Pharma is promising because Eli Lilly helps reduce risk through API support and technology transfer. Fosun’s West African plant is strategically important but must prove it can produce tablets cheaper than imports. Nigeria has the most potential, but success depends on making its own chemical ingredients and improving purchasing systems.
Natco Pharma’s recent purchase of Adcock Ingram at a valuation of 7.83x FY24 EV/EBITDA shows international confidence in South Africa as a gateway to the continent. Ultimately, while DFI funding can build the factories, only steady contracted demand, faster government approvals, and local API production will make them successful investments.